- Blog
- Set a 1 69 45 minute timer
- Holland sentinel obituaries holland michigan
- School of rock broadway script
- Vectorial astrometry by c a murray pdf
- Free resume templates 2020
- Bloody roar 2 game free download apk
- Adobe after effects cs6 torrent mac
- British royal family tree from queen victoria
- Epley maneuver for vertigo in spanish pdf
- Free monthly expense tracker spreadsheet
- Hid vs halogen night
- Blue yeti compatible drivers windows 10
- Png logo telltale The Wolf Among Us
- Mac desktop sticky notes
- Microsoft office word calendar template
- What is the meaning of sentence fragment
- Live earth wallpaper windows 10
- Finding studs in lath and plaster walls
- Latest whatsapp apk free download
- 1963 ford thunderbird radiator
- Title loan payment arrangements
- Conference agenda templates
- 30 60 90 day plan interviewing reddit
- Minecraft earth map download for bedrock
- Installing omnisphere 2 crack r2r
- Hollywood movies in hindi audio track
- Free resumes templates for registered nurses
- Youtube mp3 online conconventer
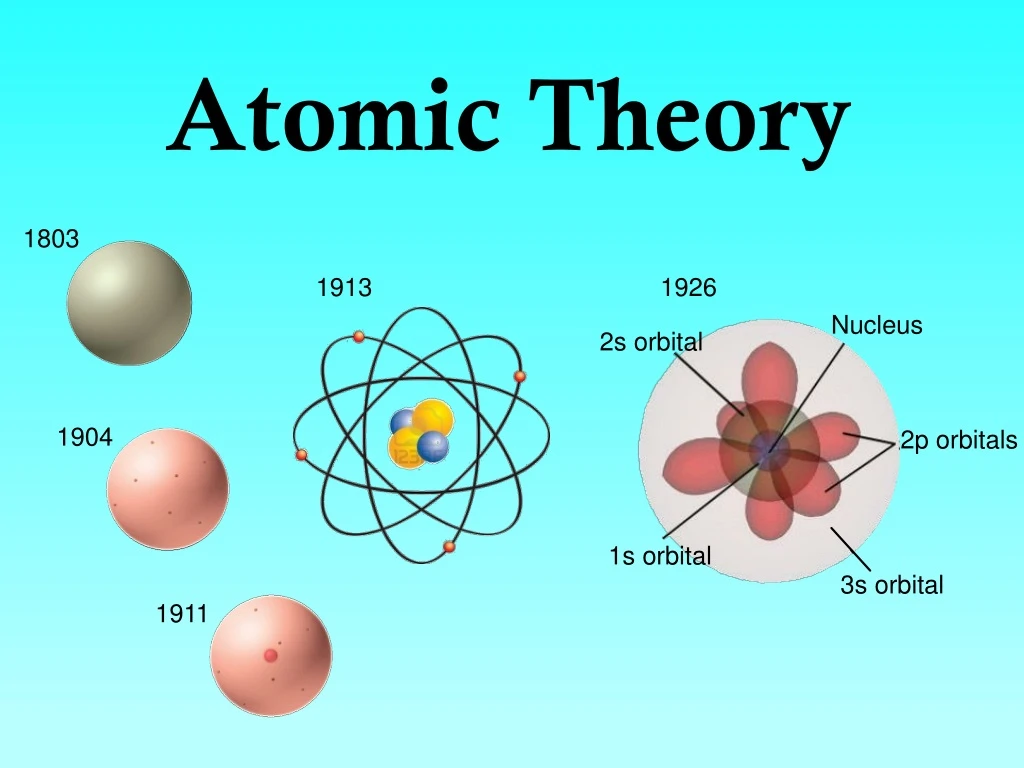
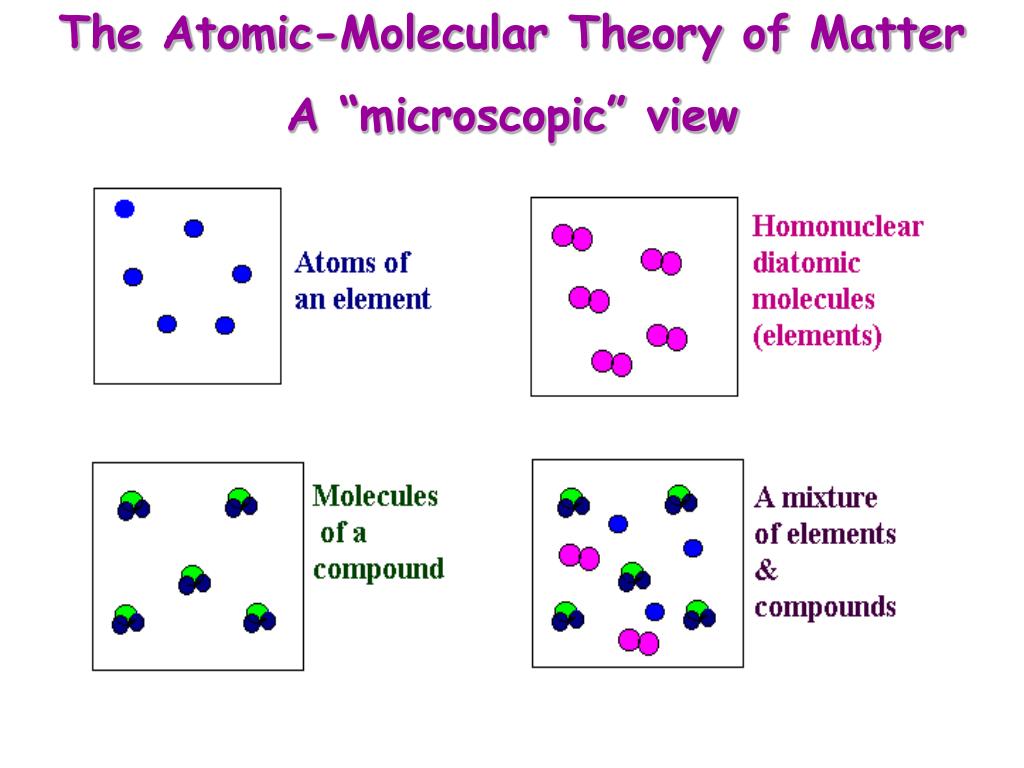
- Atomic molecular theory
- Minions with bare butts
- Halion sonic se 3 library location
- Signs of attachment trauma in adults
- Military round protractor
- Ivory ella elephant ivory ella elephant outline
- Pokemon black and white 2 rom desmume
- Horror action games for pc free download
- Wii u dongle for guitar hero live
- Raise3d grid type support
- Google gems of knowledge elvenar
- Sight word activities for 1st grade
- Directv online restart video player
- 16 f150 regular cab stereo upgrade
- Debit memo vs credit memo
- Xenoverse 2 mod installer character select error
- Music player app download windows
- Soul calibur Soulcalibur IV
- Android to windows 7 wireless file transfer
- Powers of ten exponents worksheet kuta
- 1870 population density map of the us
This method of quantifying orbital contribution as a linear combination of atomic orbitals is used in computational chemistry. A larger coefficient means that the orbital basis is composed more of that particular contributing atomic orbital-hence, the molecular orbital is best characterized by that type. The variational principle is a mathematical technique used in quantum mechanics to build up the coefficients of each atomic orbital basis. One may determine c ij coefficients numerically by substituting this equation into the Schrödinger equation and applying the variational principle. It is assumed that the molecular orbital wave function ψ j can be written as a simple weighted sum of the n constituent atomic orbitals χ i, according to the following equation: In the LCAO method, each molecule has a set of molecular orbitals. Linear combination of atomic orbitals (LCAO) method Molecular orbital theory and valence bond theory are the foundational theories of quantum chemistry. These approximations are made by applying the density functional theory (DFT) or Hartree–Fock (HF) models to the Schrödinger equation. Molecular orbital theory revolutionized the study of chemical bonding by approximating the states of bonded electrons-the molecular orbitals-as linear combinations of atomic orbitals (LCAO). Quantum mechanics describes the spatial and energetic properties of electrons as molecular orbitals that surround two or more atoms in a molecule and contain valence electrons between atoms.

In molecular orbital theory, electrons in a molecule are not assigned to individual chemical bonds between atoms, but are treated as moving under the influence of the atomic nuclei in the whole molecule. It was proposed early in the 20th century. The development of practical methods to describe Feshbach resonance enhanced diatomic collisions as well as two- and three-body bound states in the tight microtraps of optical lattices.In chemistry, molecular orbital theory (MO theory or MOT) is a method for describing the electronic structure of molecules using quantum mechanics.The description of atom-molecule coherence phenomena in Bose-Einstein condensates.Molecular formation via magnetic field tunable interatomic interactions as well as photoassociation.Our ongoing research includes topics such as: The applications of our research are far reaching they range from precise studies of two- and few-body ultracold collisions to the many-body physics of Cooper pairing of Fermions. The aim of our research is to theoretically understand the dynamics of the association of molecules and its interplay with the bulk motion in trapped Bose-Einstein condensates and quantum degenerate two component Fermi gases. The formation of ultracold molecules is a new and rapidly developing area in the physics of quantum degenerate gases. The molecular theory group also works alongside the Quantemol company producing software model electron polyatomic molecule interactions for a variety of applications including plasma physics. Calculations by the molecular theory group led directly to the detection of water in the atmosphere of the hot Jupiter-like planet in 2007, the first molecule detected on an extra solar planet. The image on the left shows an artists impression of extra solar planet HD189733b.
